Research

Cardiac Valve Form & Function
Several signalling pathways converge to orchestrate valve development and their tight temporal and spatial regulation is vital, considering the heavy load and function of valves throughout the life cycle of an organism. It is therefore not surprising that defects in valves and the associated structures septa are the most common cause of Congenital Heart Diseases (Loffredo 2000). The heart is the first organ to form and funct ion in vertebrates. In higher vertebrates, developmental abnormalities of the heart often lead to early lethality making it difficult to study later aspects of heart development such as valve formation. Zebrafish embryos develop externally, they are transparent and they survive even in the total absence of circulation until very late stages of development. All of these characteristics, together with the ease to do forward and reverse genetics, make it an ideal system to study late processes of heart morphogenesis.
In zebrafish embryos, the heart is formed at 24 hours post fertilization (hpf) as a simple, bilayer, spontaneously contracting tube: the primitive heart. The primitive heart tube consists of an outer contractile myocardium and an inner endocardium of endothelial nature. It pumps blood through a simple vascular loop. During subsequent development the heart tube loops. The two chambers of the zebrafish heart -the atrium and the ventricle- differentiate morphologically and valves form in discrete regions of the heart to prevent retrograde blood flow. Cardiac valves form at two sites along the antero-posterior axis of the developing zebrafish heart: the outflow tract and the atrio-ventricular canal. During these profound reorganizations, the heart is continuously functioning and pumping blood through a vascular network of increasing complexity. Our work (Beis et al. 2005) and work from others (Hove et al. 2003, Bartman et al. 2004, Auman et al. 2007, Scherz et al. 2008, Vermot et al. 2009) revealed the role of proper heart function in the morphogenesis of myocardium and the endocardial valves. However, the molecular mechanisms by which endocardial cells sense and transduce the shear stress and the mechanical pressure from blood flow and myocardial contractility remain elusive. The central question that arises is how morphogenetic changes and changes in function during heart development integrate. Zebrafish provides an ideal model system to study these questions in vivo (Beis and Stainier 2006). We are currently positional cloning to identify the genes from a forward genetic screen we participated in UCSF. We have identified novel alleles of myh6, GARS, PRKD2 and spaw and work on identifying the gene networks that control valve development.
In addition we are using these mutant lines for chemical genetic screens. The zebrafish embryo has become an important vertebrate model also for assessing drug effects. It is well suited for studies in genetics, embryology, development, and cell biology. Zebrafish embryos exhibit unique characteristics, including ease of maintenance and drug administration, short reproductive cycle, and transparency that permits visual assessment of developing cells and organs. Because of these advantages, zebrafish bioassays are cheaper and faster than mouse assays, and are suitable for large scale drug screening particularly for assessing toxicity, angiogenesis, and apoptosis. Increasing pharmacological data in a plethora of chemical compounds, demonstrates that toxic response, teratogenic effects, and LC50 in zebrafish are comparable to results in mice. The effects of compounds on various organs including the heart, brain, intestine, pancreas, cartilage, liver, and kidney can be observed in the transparent animals without complicated processing, demonstrating the efficiency of toxicity assays using zebrafish embryos. A key advantage of biomedical fish models is the availability of a large number of different mutant and transgenic lines.
Deciphering the Signaling Pathways that Shape a Cardiac Valve
The long-term goal of this project is to elucidate how cardiac valves form and function to support appropriate heart performance throughout the life of vertebrates. The heart is one of the first organs to form and function during vertebrate development. It starts functioning before its morphogenesis is completed.
The central question that arises is how the morphogenetic changes and the functional changes during heart development are integrated. Cardiac valves derive from endocardial cells and form along the antero-posterior axis of the vertebrate heart to prevent retrograde blood flow. Valve morphogenesis happens while the heart is contracting and blood flows through the endocardium and it is influenced by shear stress on the endothelial cells.
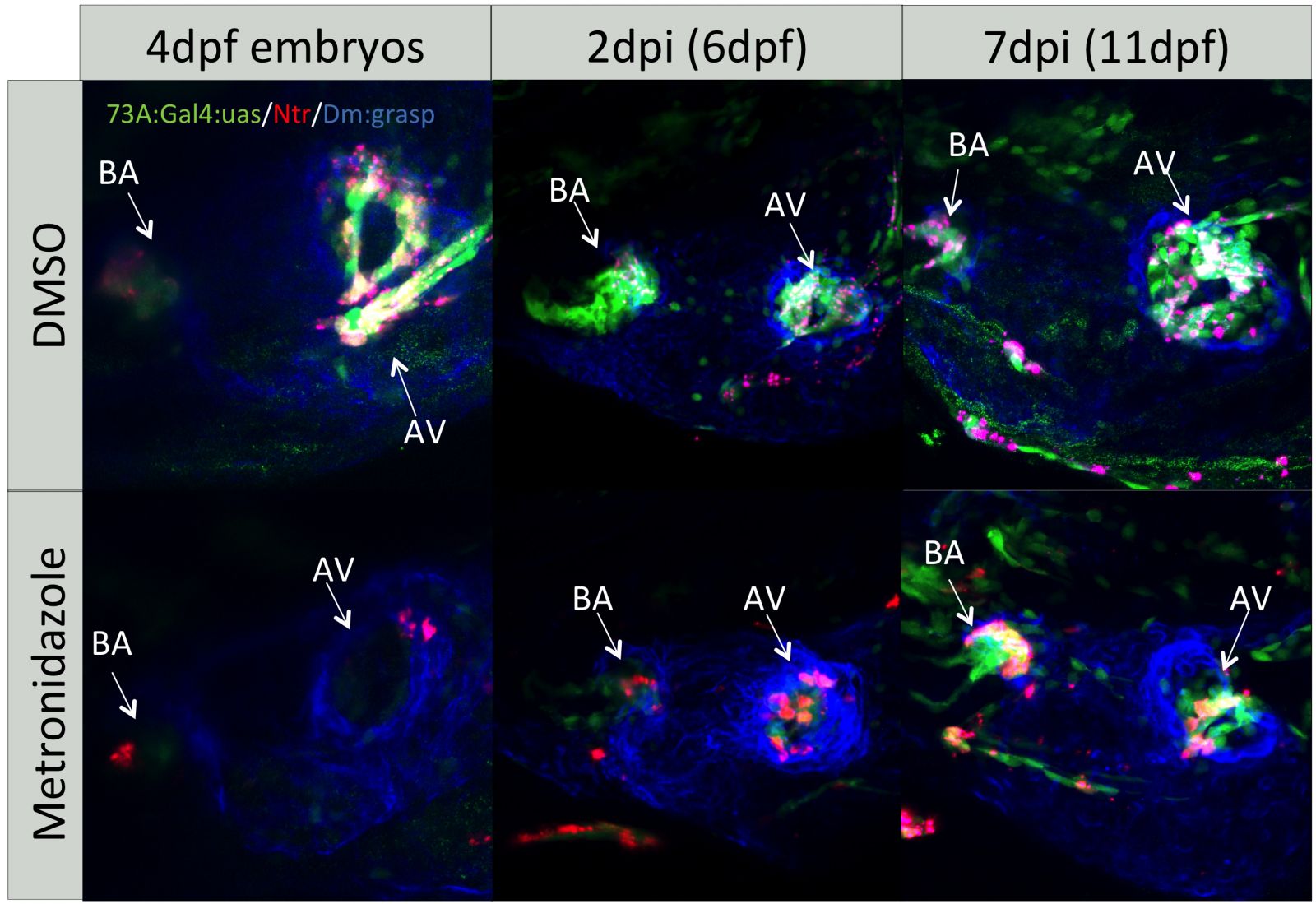
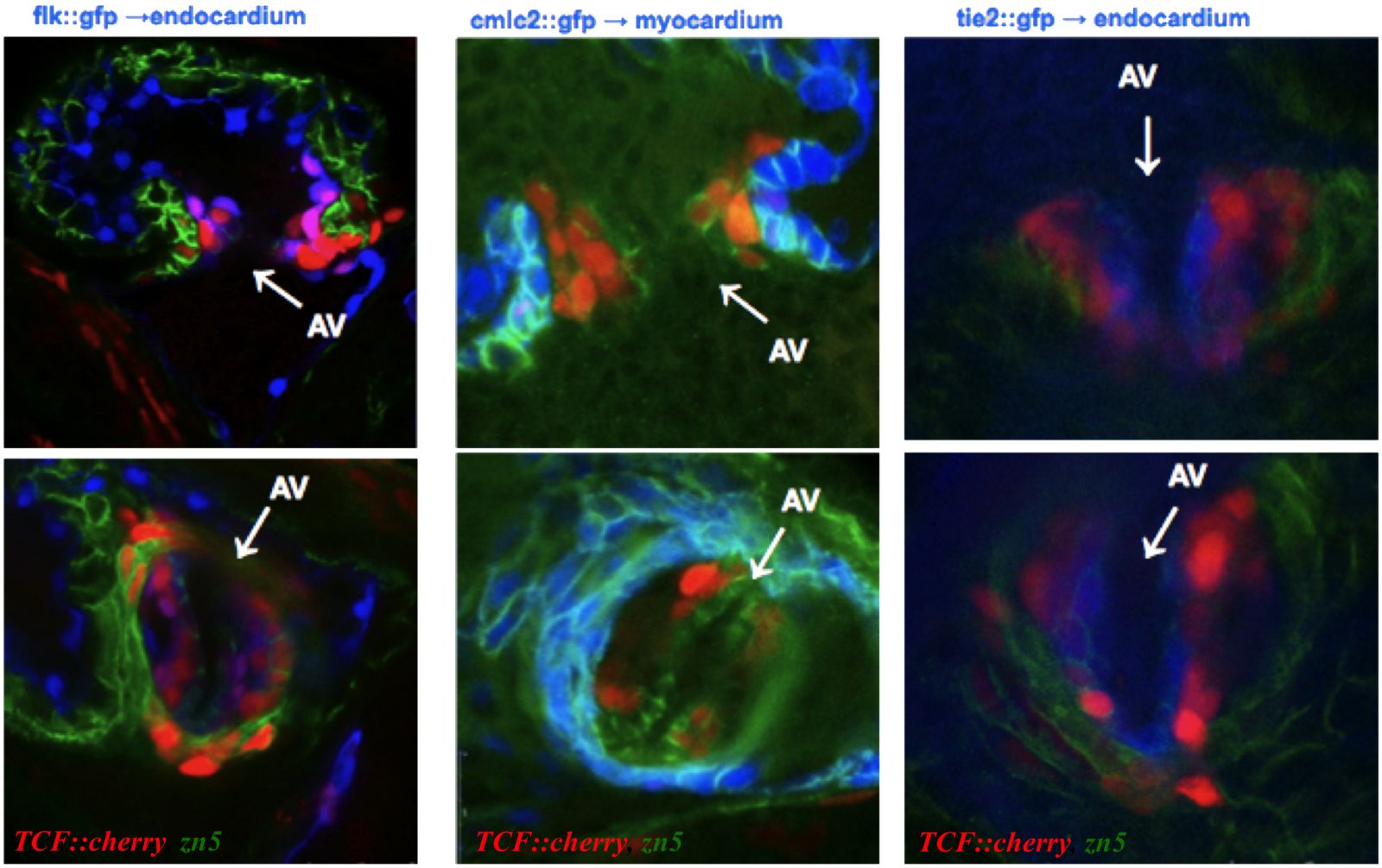
Defects in valve development affect 1% of the population and cause impaired heart function with lifelong complications. Studies in mouse and chick have been very informative in identifying key regulators and testing signaling pathways involved in valve development. However these studies are mainly in vitro and use predominantly reverse genetics approach. It is now obvious that our current knowledge will be significantly complemented by in vivo studies in zebrafish.
In order to do this, we pursue multiple lines of research using complementary experimental approaches:
-
- the cloning of the remaining of the genes in the valve mutant lines we identified in a forward genetic screen;
- elucidate the signaling pathway of protein kinase D2 in angiogenesis and valve development
- the genome-wide analyses of the endothelial shear-stress sensing mechanisms in vivo;
- the functional analyses of the novel genes by reverse genetics;
- the generation of tools to study shear-stress and endothelial mechanical sensing in vivo;
- develop the tools and a framework to study adult valve maintenance and regeneration.
We expect to develop a detailed framework of this fascinating but complex morphogenetic process which is crucial for cardiac function.

InRegen Project
Fine-tuning the inflammatory response following cardiac injury to promote cardiac regeneration
H2020-MSCA-IF-2020
Dimitris Grivas Ph.D., MC fellow
Stelios Psarras Ph.D. , Staff Research Scientist – Associate Professor Level
The pronounced inability of adult human/mammalian heart to regenerate causes millions of deaths following cardiac insult, particularly in the longer term. The extent and persistence of associated inflammation has been generally linked with adverse cardiac outcomes, including fibrosis, hypertrophy, and dysfunction. However, characteristics of the inflammatory response e.g. the maturity of resident macrophages and/or the activation status of infiltrating cells may differentially influence cardiac fibroblasts and, most importantly, cardiomyocytes, thus affecting cardiac regeneration, hypertrophy, fibrosis and dysfunction. Unraveling crucial parameters of such interactions in appropriate biological systems should confer decisive intervention potential in a serious health problem. In the past years we have studied in detail cellular and molecular players regulating pivotal events initiating or sustaining the progress to heart failure (HF) in mice and zebrafish and we propose here to combine the systems to globally study these interactions.